Allen Lab
Research in the Allen Lab
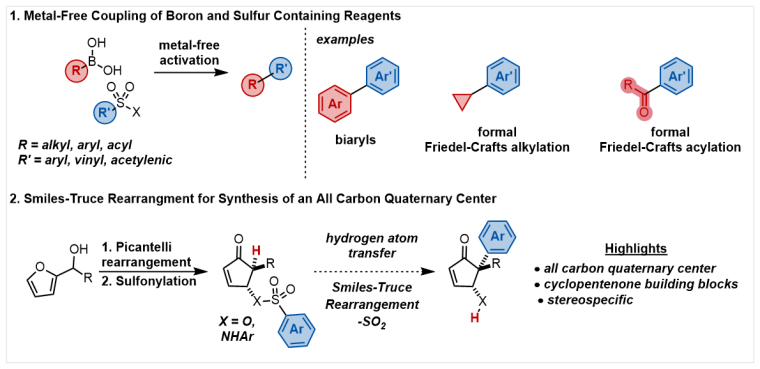
Boronic acids and sulfonyl halides are some of the most common reagents utilized in modern organic synthesis. Both classes of compounds are commercially available with diverse substitution at boron and sulfur, respectively. As such, they are ideal reagents to leverage as readily available, bench stable sources of alkyl and aryl residues. The Allen lab will investigate activation of these species to develop metal-free cross-coupling reactions.
Cyclopentenones are a privileged scaffold as building blocks in the synthesis of biologically relevant molecules. Methods that functionalize cyclopentenones will allow chemists to probe their versatility. In particular, the synthesis of all carbon α-quaternary centers in a stereospecific manner would constitute a novel advancement for the synthesis of densely functionalized cyclopentenones. Utilizing the Smiles-Truce rearrangement and hydrogen atom transfer catalysis, the Allen lab will introduce aryl groups to the α-position of cyclopentenones.
Students will learn synthetic organic and organometallic techniques including reaction set up, isolation, purification, and characterization utilizing NMR, IR, and MS. An emphasis will also be placed on student engagement with primary chemical literature and instruction in physical organic chemistry concepts & organic reaction mechanisms.
View Visiting Assistant Professor Allen's faculty bio.